State of the Art Technologies Used to Explore Long Noncoding Rnas in Cancer Disease
Abstract
The previous decade has seen long non-coding RNAs (lncRNAs) rise from obscurity to being defined as a category of genetic elements, leaving its mark on the field of cancer biological science. With the electric current number of curated lncRNAs increasing past 10,000 in the concluding 5 years, the field is moving from annotation of lncRNA expression in diverse tumours to understanding their importance in the cardinal cancer signalling networks and characteristic behaviours. Here, we summarize the previously identified too as recently discovered mechanisms of lncRNA function and their roles in the hallmarks of cancer. Furthermore, we identify novel technologies for investigation of lncRNA backdrop and their function in carcinogenesis, which will exist important for their translation to the clinic as novel biomarkers and therapeutic targets.
Background
Our understanding of cancer biology was drastically inverse by the genomic revolution of the last decade, marked by the conclusion of the human genome projection and the development of novel Dna sequencing technologies [1, 2]. The complete human genome sequence provided a framework for comparison of populations with cancer susceptibility, allowing for clinical prognosis based on mutations in genes such as BRCA1/2 or differential treatment based modifications in KRAS and BRAF [3–5]. Sequencing of private tumours revealed the prevalence of acquired DNA harm compared to the germline mutations, which allowed identification of footprints for private mutagens and gave usa important insights into tumour heterogeneity and evolution [vi–ten]. Parallel to the progress in genomics, advances in transcriptomics initiated functional note of numerous genomic loci associated with cancer that do not overlap protein-coding genes – the noncoding genome.
Large-scale cDNA sequencing projects, together with technological advancements such every bit tiling arrays and the next generation RNA sequencing provided an unprecedented view of the transcriptome complexity [xi–15]. Surprisingly, only 1–2 % of the whole genome encodes proteins, with bear witness of at least fourscore % of the remainder existence actively transcribed [11, sixteen]. These non-coding portions of the genome produce a big multifariousness of mostly regulatory RNAs that differ in their biogenesis, properties and part, and are separated past their size into curt, such as miRNAs (reviewed in [17]) and long (>200 nt) RNAs [12, eighteen–20]. The heterogeneous category of long not-coding RNAs (lncRNA) are especially abundant, accounting for 16,000 curated records in the current Gencode annotation (five.23) [21] with for all lncRNA loci in the human being genome numbering as high every bit sixty,000 [22].
lncRNAs remained elusive even in the genomics era due to their low expression levels and their presence in specific cell types, tissues or narrow time frames [23–25]. They were identified as a class of RNA molecules in 2002 [26], even though some lncRNA such equally H19 and Xist were known since the early 1990s [27, 28] Analogous to protein coding genes but with low coding potential, these RNAs are unremarkably transcribed by RNA polymerase Two (Political leader II), spliced, and mostly polyadenylated [12, thirteen]. Similarly, lncRNA promoters are enriched for active histone modifications typical of Politician II occupancy: H3K4me3, H3K9ac and H3K27ac [20, 29]. Fifty-fifty though the sequence of lncRNAs evolves rapidly, peculiarly compared to their 3D structure, their tissue specificity besides as promotor sequences remain conserved every bit poly peptide-coding genes [thirty–32]. The heterogeneity of lncRNAs resonates in the diversity of their functions; lncRNAs interact with DNA, proteins and other RNAs to participate in processes from transcription, intracellular trafficking to chromosome remodelling as reviewed previously [29, 33]).
lncRNAs have been observed to regulate complex cellular behaviours such as growth, differentiation and establishment of cell identity that are ordinarily deregulated in cancer [34–36]. Some have already been linked to poor prognosis in multiple neoplasm types and have a clinical relevance as biomarkers. In this review we will focus on the molecular mechanisms of part for cancer-associated lncRNAs, their involvement in cancer hallmarks and provide data on the most recent advances in technologies for their identification and functional interrogation.
Identification of lncRNAs in cancer
lncRNAs were initially observed in carcinogenesis due to their differential expression compared to normal tissues. High expression in tumour tissues of some of the kickoff identified lncRNAs such every bit h19, MALAT1 and PCA3 was recognised earlier the availability of side by side-generation sequencing technologies [37–39]. RNA sequencing allowed a large-scale cess of differential expression of lncRNAs comparing cancer to normal tissues, with a large number of lncRNAs showing abnormal expression, similar to the influence microarrays had on the miRNA field (Fig. 1). Recently, a number of lncRNAs have been systematically identified in numerous cancer transcriptomes, either by overlap of sequencing libraries with previously annotated GENCODE lncRNAs, or by de novo assembly of all available public datasets [22, 40], marking their presence in the majority of cancer types (Fig. ii, Additional file 1: Tabular array S1). Novel RNA-seq techniques such as CaptureSeq, that enriches transcript libraries for specific oligonucleotides designed for genomic regions of involvement, volition improve observation of rare or lowly abundant transcripts from gene deserts associated with cancer [41].
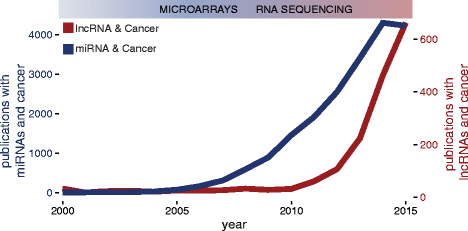
Interest for lncRNAs (reddish) in the cancer scientific community compared to miRNAs (blue). The y-axis represents the number of publications and the x-axis represents time. Data was obtained by searching Pubmed for 'lncRNA cancer' or 'miRNA cancer'. Data from 2022 was not used in the graph. Publications with terms 'miRNA' and 'cancer' plateau in 2015
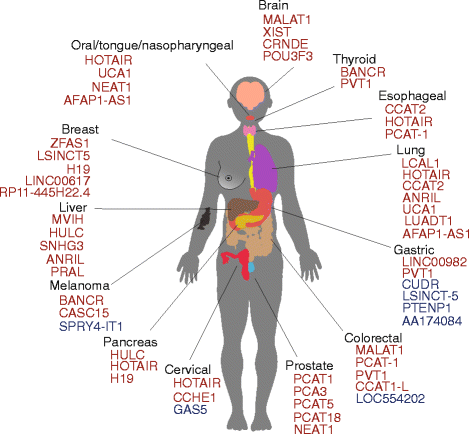
Some lncRNAs associated with cancers. The colour represents either upregulated (red) or downregulated (bluish) compared to normal tissues
In that location are ii main drivers for altered expression of lncRNAs in tumours. Beginning, abundance of some lncRNAs can be contradistinct due to the cancer-induced alter of re-create number of their genomic loci. This miracle has been observed for FAL1 in ovarian and PRAL in hepatocellular carcinoma [42, 43]. Second, expression of some cancer-associated lncRNAs tin can be initiated by oncogenes interim as transcription factors. Ii crucial genes implicated in multiple tumour types, Myc and p53, act as transcription factors for a large number of lncRNAs [44–46]. Some of these lncRNAs modulate activity of their respective TFs in a feedback loop, for example MINCR with Myc and MEG3 for p53, which makes them potential candidates for therapeutic targeting [47, 48].
Presence of lncRNAs in specific tumours tin can also be observed based on their overlap with the cancer risk loci identified through genome-broad association studies (GWAS). For example, clan of ANRIL with glioma and basal prison cell carcinoma likewise as PTCSC3 with thyroid cancer were discovered based on the known take chances loci established through genotyping of cancer patients [49, 50] Recently, CASC15 and NBAT1 were identified through GWAS of neuroblastoma, a cancer that mostly affects children and has a poorly explained genetic background. Both CASC15 and NBAT1 are role of the 6p22 locus that contains SNP rs6939340, associated with metastatic illness and poor event free survival [51, 52]. CASC15 acts as a neoplasm suppressor and is associated with advanced tumour stages and poor patient survival, while NBAT1 seems to negatively regulate transcription factor NRSF (neuron restrictive silencing factor) [52, 53]. Furthermore, CARLo-5 was identified as significantly correlated with the rs6983267 allele, a single nucleotide polymorphism (SNP) in the region for Myc enhancers. This lncRNA is associated with increased cancer susceptibility and has a function in cell-cycle regulation and tumour development [54]. Since more than 80 % of disease associated SNPs fall into intronic and intergenic regions, it is likely that time to come development of more than sensitive technologies for RNA detection will be essential in defining novel cancer-associated lncRNAs [55].
Mechanisms of lncRNA function in carcinogenesis
Although lncRNAs are scarcely functionally annotated [56], their machinery of action can exist separated based on their influence on chromatin state and methylation, stability of proteins and complexes or by acting as a sponge for miRNA inhibition (reviewed in [29]).
Chromatin land and methylation
Chromatin remodelling was i of the commencement identified functions of lncRNAs. Epigenetic remodelling is frequently accomplished through interaction of a lncRNAs with PRC2 (reviewed in [57]), a protein from the polycomb complex that introduces chromatin inactivation past establishing inhibitory H3K427me3 histone marks [58]. These lncRNAs are sometimes expressed antisense to the target gene involved in cancer, such every bit in the case of ANRIL with CDKNA/B and asFGFR2 with FGFR2 [59, lx], suggesting a possibility for a cis-acting activity. PCE3, as the first lncRNA involved in cancer was simply recently found to accept the same mode of activeness, with the discovery of its antisense protein oncogene PRUNE2 [61]. However, it has also been observed that PRC2 lacks specificity in binding RNAs that recruit it, providing a potential caption why such a big number of lncRNAs influence chromatin remodelling [62]. Besides acting through PRC2, some lncRNAs such as Kcnq1ot1, TARID, AS1DHRS4 and DACOR1 recruit Dna methyltransferases directly to modify chromatin conformation, or they alter nucleosome positioning through SWI/SNF complex equally in the case of SChLAP1 [63–67]. Other lncRNAs, similar Firre, bind chromatin remodelers cohesin and CTCF to change chromatin country of the whole chromosomes in the process of X chromosome inactivation [68]. lncRNAs can also human action every bit chromatin activators, as in the case of HOTTIP and CCAT1-L that regulate chromosome looping in their proximity to eolith activating H3K4me3 marker on gene promoters [69–71].
Stability of proteins or protein complexes
A large number of lncRNAs exert their oncogenic part through straight interaction with proteins or protein complexes as scaffolds or allosteric activators/inhibitors. CTBP1-AS, CCTA2 and ZBTB7A interact with transcription factors and modify their activity [72–74]. Some lncRNAs tin exist used as a scaffold for assembly of whole protein complexes, for example HOTAIR for the HBXIP/Hotair/LSD1 complex [75], NEAT1 for paraspeckle proteins [76, 77], BCAR4 for binding of SNIP1 and PNUTS [78], PRAL for HSP90 and p53 [43]. Furthermore, TERRA recruits a circuitous of chromatin modifiers to regulate telomere maintenance in response to a variety of cellular signals [79]. Finally, some lncRNAs such as GAS5 bind to nuclear receptors, in this case the glucocorticoid receptor [80].
Competing endogenous RNAs
Some lncRNAs have recently been found to deed as competing endogenous RNAs (ceRNAs), by bounden miRNAs ("sponging") and reducing their inhibitory effect on their natural targets (reviewed in [81]). There are numerous examples of lncRNA sponges involved in cancer progression. In lung cancer, UCA1 up-regulates a stiff oncogene ERBB4 by binding miR-193-3p [82]. In gastric cancer, MEG3 upregulates Bcl-ii by sequestering miR-181-a [83]. Similarly, ZFAS1 binds miR-150 in hepatocellular carcinoma, Linc-RoR binds miR-145 in endometrial cancer stem cells and CASC2 regulates concentration of miR-21 [84–86].
In addition to the previously described mechanisms, lncRNAs have recently been observed to employ the following fix of diverse strategies. lncRNAs tin can alter the phosphorylation state of proteins by masking phosphorylation motifs, like LINK-a and HIF1a [87]. Furthermore, some lncRNAs such every bit NORAD act as sponges for a whole set of proteins, in this instance the PUMILIO family that would otherwise drive chromosomal instability past repressing mitotic, DNA repair, and DNA replication factors [88]. Interestingly, some lncRNAs grade DNA-RNA triplexes that regulate expression of oncogenes either in cis for Khps1, or in trans for MEG3 [89, ninety]. Other lncRNAs such as Uc.283 + A control production of miRNAs by influencing processing of the pri-miRNA transcripts, in this example pri-miR-195 [91].
In summary, lncRNAs human action through an increasingly broad range of mechanisms that compete with proteins in terms of their multifariousness and regulatory potential. In the adjacent section we discuss how these mechanisms bear on upon prison cell transformation to cancer phenotype.
Hallmarks of cancer
In 2000, Hanahan and Weinberg described six properties required for cell transformation, termed hallmarks of cancer. These included cocky-sustained growth signalling, insensitivity to growth inhibition, apoptosis avoidance, uncontrolled proliferation, angiogenesis and metastasis [92, 93]. lncRNAs as regulatory molecules have been implicated in the majority of these functions (reviewed in [94]), and key patterns are starting to sally.
Cocky-sustained growth signalling
lncRNAs promote self-sufficiency in growth signals generally by interim on the activation on the commencement step of the point transduction, the signal receptors. Multiple lncRNAs accept been observed to specifically demark nuclear receptors either alone or in a ribonucleoprotein complexes (reviewed in [95]). SRA1 serves as a scaffold to stabilize the estrogen receptor [96, 97], while GAS5 acts as the competitive inhibitor of the glucocorticoid receptor [80]. Instead of activating the point receptors, some lncRNAs such as PVT1 affect the proliferation by regulating the receptor abundance, as is demonstrated for PVT1 and thyroid-stimulating hormone [98].
Insensitivity to growth inhibition
Growth inhibition or its evasion can also be regulated by lncRNAs, and generally involves influence on neoplasm suppressors that regulate cell cycle such as cyclins, CDKs, CDK inhibitors and p53 (reviewed in [99]). This can be accomplished by chromatin repression through Cathay complex, as detailed in the previous section. Using this mechanism PANDA represses protein CDKN1A through PRC1 while ANRIL repress their targets tumour suppressor p15 (CDKN2B) through PRC2 [59, 100, 101]. Some lncRNAs regulate expression of neoplasm suppressors past influencing various parts of transcription and translation. Initiation of transcription can be influenced by scaffolding of transcription factor complexes, as in the case of LincRNA-p21 and p21 (inhibitor of CDK2) [102]. Transcription elongation tin can be modified by destabilization of mRNA transcripts, every bit exemplified by gadd7 and Cdk6 [103]. Finally, transcript stability and translation can be regulated postal service-transcriptionally by diminishing the part of miRNAs, every bit in the case of PTENP1 acting as a competitive endogenous RNA to inhibit miRNA repression of PTEN [104]. For some of the lncRNAs like CASC15-S, the direct mechanism of growth inhibition is unknown, just the lack of its expression increases cancer growth and migratory capacity [53].
Avoiding apoptosis
Apoptosis or controlled jail cell death is one of the primal pathways for control of carcinogenesis (reviewed in [36]). Some lncRNAs act on regulation of transcription of fundamental apoptotic genes. For example, lncRNA INXS is expressed from the intron of BCL-X and regulates its splicing into a pro-apoptotic isoform BCL-XS [105]. A recently discovered lncRNA PRAL induces apoptosis past stabilizing a circuitous between HSP90 and p53 [43]. Several other lncRNAs have been implicated in apoptosis such every bit SPRY4-IT1 [106], HOXA-AS2 [107] and uc002mbe.2 [108], but the details of the machinery of action remain still unknown.
Uncontrolled proliferation
Maintenance of telomeres as nucleoprotein structures that stabilize ends of chromosomes is a primal requirement for limitless replicative potential of cancer cells. Telomeres shorten in dividing cells, then a ribonucleoprotein complex telomerase is required to elongate telomeric repeats through reverse transcription of an internal template RNA. Shortening of telomeres induces production of a lncRNAs TERRA, that is transcribed from the subtelomeric regions [109]. Nether normal conditions, TERRA represses its expression through chromatin modifications, but when activated can recruits poly peptide complexes for homology-directed repair of shortened or damaged telomeric sequences [79].
Promotion of angiogenesis
Multiple lncRNAs accept been implicated in regulation of nutrient supply to tumours, mostly by regulating vascular endothelial growth factor (VEGF) that is essential for germination of claret vesicles. Transcription of VEGF was recently reported to be regulated by lncRNAs HOTAIR [110] and MIAT [111]. MIAT sequesters mir-150-5p that is required for repression of VEGF, resulting in microvascular dysfunction and decreased metastasis after MIAT knockdown. MVIH also influences production of VEGF, though indirectly through phosphoglycerate kinase 1 (PGK1) [112]. Finally, MALAT1 has been observed to promotes angiogenic sprouting and migration when expressed in endothelial cells [113].
Tissue invasion and metastasis
Multiple lncRNAs increase invasiveness of cancer cells and facilitate metastasis. Examples of these include h19 [114], MALAT1 in colorectal and nasopharyngeal carcinoma [115], SPRY4-IT1 in melanoma [106], HOTAIR [116], AFAP1-AS1 [117], and CCAT2 [118] in lung cancer, lincRNA-RoR in breast cancer [119], LEIGC in gastric cancer [120] and lncRNA-ATB in hepatocellular carcinoma [121]. Out of these, only lincRNA-RoR and lncRNAs-ATB accept a suggested mechanism of activeness in tissue invasion. lincRNA-RoR likely serves as a "sponge" for miR-145 that is important for regulation of ADP-ribosylation factor 6, a protein involved in invasion of chest cancer cells [119]. Similarly, lncRNA-ATB, acts as a ceRNA to reduce the consequence of the miR-200 family targets ZEB1 and ZEB2, two transcription factors that promote jail cell move and metastasis [121].
lncRNAs tin exist involved in a number of other processes related to cancer. Some lncRNAs promote a metabolic switch to glycolysis and lactic acid fermentation termed the Warburg upshot [122]. lincRNA-p21 regulates the Warburg effect by preventing ubiquitination of hypoxia-inducible cistron-1 (HIF-i), a key transcription factor that promotes upregulation of glycolysis and downregulation of oxidative phosphorylation [123]. Several lncRNAs have been observed as essential for Dna repair past homologous recombination (HR): ANRIL, PCAT1 and DDSR1. Although the mechanism of ANRIL in Hr remains unknown, PCAT1 posttranslationally inhibits BRCA2 [124], while DDSR1 is suggested to collaborate with BRCA1 [125]. Finally, there are implications of lncRNAs on cancer therapies through expression of drug exporters. For case, MRUL promotes expression of ABCB1 that is essential for multidrug-resistance in gastric cancer cell lines [126].
Novel techniques for lncRNA interrogation
The number of annotated transcribed genomic elements has increased by 100 % in the last decade, the majority of which are in the non-coding space and take a defined function in less than 1 % of cases [56]. Such a vast number of novel genetic players presents a great potential for clinical applications, especially in view of cancer as a genomic disease. However, it also requires a thorough rethinking of our basic premises on biological systems, pathway structure and information transfer, every bit well every bit a articulate technological strategy to identification of their function.
The first challenge is presented by the lack of an exhaustive definition of the total cancer transcriptome, regardless of the jail cell or tissue type. Currently, a major obstacle to analysis of cancer transcriptome is the alignment of sequence reads to the consensus human genome. Ideally, all the reads would be aligned to a genome sequenced past single-molecule Deoxyribonucleic acid sequencing, but the cost and the quality of this engineering are still keeping it abroad from mainstream research. The next issue is the limited dynamic range of transcript detection for RNA sequencing. This tin already be solved by applying the recently developed CaptureSeq method for targeted enrichment of transcripts from specific regions of interest [41]. Furthermore, long read sequencing will be essential for discovery of lncRNAs isoforms and novel exons [127]. In combination with single cell sequencing it will permit identification of individual lncRNAs species from cancer subpopulations, avoiding the heterogeneity of tissue mixture.
Later on defining the non-coding elements of the transcriptome, the second challenge is the systematic identification of lncRNAs backdrop that could atomic number 82 to identifying their cellular function. This tin be achieved by investigating their location in the cellular compartments, structural properties too every bit possible interactors.
Quantified localization of lncRNAs through microscopy techniques can provide important information about their properties. RNA-Fish equally an established technique for RNA localisation has recently been used to identify subcellular location of multiple lncRNAs, in add-on to their expression across a population of cells, spatio-temporal behaviour and coexpression with proximal mRNAs [128].
Structure of biological molecules is vital to their part, and several techniques have been adult to investigate secondary and 3rd structures of lncRNAs. Techniques such every bit Parallel Analysis of RNA Structure (PARS) [129] and Fragmentation Sequencing (FragSeq) [130] sequence RNAs after specific cleavage of unmarried (FragSeq) or single and double stranded (PARS) nucleic acids, allowing for identification of loops in RNA-structure. Another way to investigate structure is to tag the flexible two′-hydroxyl groups in the RNA backbone by Selective ii′-hydroxyl Acylation and Primer Extension (SHAPE) [131]. Finally, similarly to DNA, RNA can exist edited with chemical modifications that modify its structure and binding properties. Two established methods tin can be used to place methylated RNA sites: Methylated RNA Immunoprecipitation with next-generation sequencing (MeRIP) [132], or its adaption for hydroxymethylcytosine sites – hMeRIP [133]. Another common RNA-modification is chemical modify of nucleotides adenosin to inosin, which can be detected by inosine chemical erasing sequencing (Ice-seq) [134].
Assessing the function of lncRNAs by identifying their binding partners tin be performed depending on the type of interaction. Binding of RNA to DNA or proteins tin exist assessed with ChIRP-seq or ChIRP-MS respectively (Chromatin Isolation past RNA purification followed by sequencing or mass spectrometry) [135, 136]. The specificity of ChIRP is guaranteed by pick of merely those RNA that are bound by biotinylated oligonucleotides, like to RAP [137] and Chart [138], as well as past crosslinking of RNA with Dna or protein by UV or formalin. A contempo modification to the protocol tin can observe individual RNA domains that collaborate with DNA, RNA or proteins [139]. Instead of biotinylated oligonucleotides, RNA-guided chromatin conformation capture (R3C) reverse-transcribes RNA bound to DNA into cDNA with biotin labelling and joins it with the adjacent genomic Deoxyribonucleic acid with T4 DNA ligase, allowing for streptavidin selection and sequencing [140]. Furthermore, identification of lncRNAs that bind to a protein of involvement such equally PRC2 [141] tin can be performed through RNA Immunoprecipitation [142] that was later on coupled with sequencing (RIP-Seq) [65]. The specificity of RIP has been improved in by UV crosslinking of RNA and protein in Cross-Linking ImmunoPrecipitation (Clip) [143] and the later modifications with sequencing (HITS-CLIP) [144] and iClip [145]. Finally, the affinity of a protein for multiple RNA can be assessed in a high-throughput manner. This tin can be achieved either on a microfluidic platform past RNA-mechanically Induced Trapping of Molecular Interactions (RNA-MITOMI) [146], or on a flow cell in RNA-MaP (massively parallel assortment) [147].
The terminal challenge in defining lncRNA functions is developing loss- and gain-of-function lncRNA studies. The RNA interference technology is being supplemented by the powerful CRISPR/Cas-9 system, a newly developed genome-editing applied science that allows easier manipulation of lncRNAs behaviour [148]. CRISPR allows multiple types of manipulation, from deletion of diverse parts of genomic lncRNAs loci, to insertion of promoters, and novel exons. A recent modification of the CRISPR technique that was developed in Rinn grouping allows insertion of RNA domains to genomic loci, allowing for identification of in cis behaviour of lncRNAs [149].
Determination
Long not-coding RNAs are fine-tuners and regulators of primal biological processes. Though nosotros have only started to annotate their function in various aspects of cell transformation and metastasis, they are already filling in the major gaps of our understanding of cancer biological science. It will be exciting to see the side by side decade migrate from the perception of lncRNAs equally a side act in biological regulation to the center of new biological concepts, paradigms and drug therapies. Watch this infinite.
Abbreviations
ceRNA, competing endogenous RNAs; Nautical chart, capture hybridization of analysis of RNA targets; ChIRP-seq or ChIRP-MS, chromatin isolation by RNA purification followed by sequencing or mass spectrometry; Prune-seq, cantankerous-linking immunoprecipitation sequencing; GWAS, genome-broad association studies; hMeRIP, hydroxymethylcytosine sites in RNA; lncRNA, long noncoding RNA; MeRIP, methylation RIP-seq; PARS, parallel analysis of RNA structure; RAP, RNA antisense purification; RIP, RNA immunoprecipitation; RNA-MaP, RNA on a massively parallel array RNA-MITOMI, RNA-mechanically induced trapping of molecular interactions; SHAPE, selective 2′-hydroxyl acylation and primer extension; SNP, single nucleotide polymorphism
References
-
Wheeler DA, Wang L. From man genome to cancer genome: the first decade. Genome Res. 2013;23:1054–62.
-
Offit Grand. Decade in review--genomics: a decade of discovery in cancer genomics. Nat Rev Clin Oncol. 2014;11:632–4.
-
Pal T, Permuth-Wey J, Betts JA, Krischer JP, Fiorica J, Arango H, LaPolla J, Hoffman M, Martino MA, Wakeley Thou, Wilbanks G, Nicosia S, Cantor A, Sutphen R. BRCA1 and BRCA2 mutations business relationship for a big proportion of ovarian carcinoma cases. Cancer. 2005;104:2807–16.
-
Lievre A, Bachet J-B, Le Corre D, Boige V, Landi B, Emile J-F, Cote J-F, Tomasic G, Penna C, Ducreux M, Rougier P, Penault-Llorca F, Laurent-Puig P. KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res. 2006;66:3992–v.
-
Zlobec I, Bihl MP, Schwarb H, Terracciano L, Lugli A. Clinicopathological and poly peptide characterization of BRAF- and Thou-RAS-mutated colorectal cancer and implications for prognosis. Int J Cancer. 2010;127:367–80.
-
Forbes SA, Beare D, Gunasekaran P, Leung K, Bindal North, Boutselakis H, Ding G, Bamford S, Cole C, Ward Due south, Kok CY, Jia 1000, De T, Teague JW, Stratton MR, McDermott U, Campbell PJ. Cosmic: exploring the globe'southward noesis of somatic mutations in man cancer. Nucleic Acids Res. 2015;43(Database event):D805–eleven.
-
Alexandrov LB, Nik-Zainal S, Wedge DC, Aparicio SAJR, Behjati S, Biankin AV, Bignell GR, Bolli Northward, Borg A, Børresen-Dale A-50, Boyault S, Burkhardt B, Butler AP, Caldas C, Davies HR, Desmedt C, Eils R, Eyfjörd JE, Foekens JA, Greaves M, Hosoda F, Hutter B, Ilicic T, Imbeaud S, Imielinski M, Jager N, Jones DTW, Jones D, Knappskog S, Kool M, et al. Signatures of mutational processes in human cancer. Nature. 2013;500:415–21.
-
Campbell PJ, Pleasance ED, Stephens PJ, Dicks E, Rance R, Goodhead I, Follows GA, Dark-green AR, Futreal PA, Stratton MR. Subclonal phylogenetic structures in cancer revealed by ultra-deep sequencing. Proc Natl Acad Sci U Due south A. 2008;105:13081–6.
-
Lawrence MS, Stojanov P, Polak P, Kryukov GV, Cibulskis K, Sivachenko A, Carter SL, Stewart C, Mermel CH, Roberts SA, Kiezun A, Hammerman PS, McKenna A, Drier Y, Zou L, Ramos AH, Pugh TJ, Stransky Due north, Helman Eastward, Kim J, Sougnez C, Ambrogio L, Nickerson Eastward, Shefler Due east, Cortés ML, Auclair D, Saksena G, Voet D, Noble M, DiCara D, et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature. 2013;499:214–8.
-
Navin N, Kendall J, Troge J, Andrews P, Rodgers L, McIndoo J, Cook K, Stepansky A, Levy D, Esposito D, Muthuswamy L, Krasnitz A, McCombie WR, Hicks J, Wigler M. Tumour evolution inferred by unmarried-prison cell sequencing. Nature. 2011;472:xc–4.
-
Consortium EP. An integrated encyclopedia of DNA elements in the human genome. Nature. 2012;489:57–74.
-
Carninci P, Kasukawa T, Katayama Southward, Gough J, Frith MC, Maeda N, Oyama R, Ravasi T, Lenhard B, Wells C, Kodzius R, Shimokawa Thou, Bajic VB, Brenner SE, Batalov Due south, Forrest ARR, Zavolan Thou, Davis MJ, Wilming LG, Aidinis Five, Allen JE, Ambesi-Impiombato A, Apweiler R, Aturaliya RN, Bailey TL, Bansal 1000, Baxter 50, Beisel KW, Bersano T, Bono H, et al. The transcriptional landscape of the mammalian genome. Scientific discipline. 2005;309:1559–63.
-
Bertone P, Stolc 5, Royce TE, Rozowsky JS, Urban AE, Zhu X, Rinn JL, Tongprasit W, Samanta Yard, Weissman S, Gerstein Yard, Snyder M. Global identification of human transcribed sequences with genome tiling arrays. Science. 2004;306:2242–6.
-
Dinger ME, Amaral PP, Mercer TR, Mattick JS. Pervasive transcription of the eukaryotic genome: functional indices and conceptual implications. Cursory Funct Genomic Proteomic. 2009;8:407–23.
-
Clark MB, Amaral PP, Schlesinger FJ, Dinger ME, Taft RJ, Rinn JL, Ponting CP, Stadler PF, Morris KV, Morillon A, Rozowsky JS, Gerstein MB, Wahlestedt C, Hayashizaki Y, Carninci P, Gingeras TR, Mattick JS. The reality of pervasive transcription. PLoS Biol. 2011;nine:e1000625. discussion e1001102.
-
Hangauer MJ, Vaughn IW, McManus MT. Pervasive transcription of the human genome produces thousands of previously unidentified long intergenic noncoding RNAs. PLoS Genet. 2013;9:e1003569.
-
Lin S, Gregory RI. MicroRNA biogenesis pathways in cancer. Nat Rev Cancer. 2015;15:321–33.
-
Engstrom PG, Suzuki H, Ninomiya N, Akalin A, Sessa L, Lavorgna Chiliad, Brozzi A, Luzi 50, Tan SL, Yang L, Kunarso Thousand, Ng EL-C, Batalov S, Wahlestedt C, Kai C, Kawai J, Carninci P, Hayashizaki Y, Wells C, Bajic VB, Orlando V, Reid JF, Lenhard B, Lipovich L. Complex Loci in human and mouse genomes. PLoS Genet. 2006;2:e47.
-
Kapranov P, Cheng J, Dike South, Nix DA, Duttagupta R, Willingham AT, Stadler PF, Hertel J, Hackermuller J, Hofacker IL, Bong I, Cheung E, Drenkow J, Dumais E, Patel S, Helt G, Ganesh K, Ghosh South, Piccolboni A, Sementchenko 5, Tammana H, Gingeras TR. RNA maps reveal new RNA classes and a possible function for pervasive transcription. Science. 2007;316:1484–8.
-
Guttman M, Amit I, Garber Thou, French C, Lin MF, Feldser D, Huarte M, Zuk O, Carey BW, Cassady JP, Cabili MN, Jaenisch R, Mikkelsen TS, Jacks T, Hacohen Due north, Bernstein BE, Kellis M, Regev A, Rinn JL, Lander ES. Chromatin signature reveals over a thousand highly conserved large non-coding RNAs in mammals. Nature. 2009;458:223–7.
-
Harrow J, Frankish A, Gonzalez JM, Tapanari E, Diekhans M, Kokocinski F, Aken BL, Barrell D, Zadissa A, Searle South, Barnes I, Bignell A, Boychenko V, Hunt T, Kay Chiliad, Mukherjee G, Rajan J, Despacio-Reyes G, Saunders Grand, Steward C, Harte R, Lin M, Howald C, Tanzer A, Derrien T, Chrast J, Walters N, Balasubramanian Southward, Pei B, Tress 1000, et al. GENCODE: the reference homo genome annotation for The ENCODE Project. Genome Res. 2012;22:1760–74.
-
Iyer MK, Niknafs YS, Malik R, Singhal U, Sahu A, Hosono Y, Barrette TR, Prensner JR, Evans JR, Zhao Southward, Poliakov A, Cao X, Dhanasekaran SM, Wu Y-Grand, Robinson DR, Beer DG, Feng FY, Iyer HK, Chinnaiyan AM. The landscape of long noncoding RNAs in the human transcriptome. Nat Genet. 2015;47:199–208.
-
Mercer TR, Dinger ME, Sunkin SM, Mehler MF, Mattick JS. Specific expression of long noncoding RNAs in the mouse brain. Proc Natl Acad Sci U Southward A. 2008;105:716–21.
-
Cabili MN, Trapnell C, Goff Fifty, Koziol M, Tazon-Vega B, Regev A, Rinn JL. Integrative note of human large intergenic noncoding RNAs reveals global backdrop and specific subclasses. Genes Dev. 2011;25:1915–27.
-
Gloss BS, Dinger ME. The specificity of long noncoding RNA expression. Biochim Biophys Acta. 1859;2016:16–22.
-
Okazaki Y, Furuno G, Kasukawa T, Adachi J, Bono H, Kondo Southward, Nikaido I, Osato N, Saito R, Suzuki H, Yamanaka I, Kiyosawa H, Yagi G, Tomaru Y, Hasegawa Y, Nogami A, Schonbach C, Gojobori T, Baldarelli R, Colina DP, Bult C, Hume DA, Quackenbush J, Schriml LM, Kanapin A, Matsuda H, Batalov S, Beisel KW, Blake JA, Bradt D, et al. Assay of the mouse transcriptome based on functional annotation of threescore,770 full-length cDNAs. Nat Genet. 2002;420:563–73.
-
Brannan CI, Dees EC, Ingram RS, Tilghman SM. The product of the H19 cistron may part as an RNA. Mol Cell Biol. 1990;10:28–36.
-
Brockdorff Northward, Ashworth A, Kay GF, McCabe VM, Norris DP, Cooper PJ, Swift South, Rastan Due south. The product of the mouse Xist gene is a 15 kb inactive Ten-specific transcript containing no conserved ORF and located in the nucleus. Cell. 1992;71:515–26.
-
Quinn JJ, Chang HY. Unique features of long non-coding RNA biogenesis and part. Nat Rev Genet. 2015;17:47–62.
-
Smith MA, Gesell T, Stadler PF, Mattick JS. Widespread purifying selection on RNA structure in mammals. Nucleic Acids Res. 2013;41:8220–36.
-
Rands CM, Meader S, Ponting CP, Lunter One thousand. 8.2 % of the human genome is constrained: variation in rates of turnover across functional element classes in the human lineage. PLoS Genet. 2014;10:e1004525.
-
Necsulea A, Soumillon One thousand, Warnefors 1000, Liechti A, Daish T, Zeller U, Baker JC, Grutzner F, Kaessmann H. The evolution of lncRNA repertoires and expression patterns in tetrapods. Nature. 2014;505:635.
-
Rinn JL, Chang HY. Genome regulation by long noncoding RNAs. Annu Rev Biochem. 2012;81:145–66.
-
Hu W, Alvarez-Dominguez JR, Lodish HF. Regulation of mammalian cell differentiation past long non-coding RNAs. EMBO Rep. 2012;thirteen:971–83.
-
Flynn RA, Chang HY. Long noncoding RNAs in prison cell-fate programming and reprogramming. Cell Stalk Cell. 2014;14:752–61.
-
Rossi MN, Antonangeli F. LncRNAs: new players in apoptosis control. Int J Cell Biol. 2014;2014:473857.
-
Zhang Y, Shields T, Crenshaw T, Hao Y, Moulton T, Tycko B. Imprinting of human H19: allele-specific CpG methylation, loss of the active allele in Wilms tumor, and potential for somatic allele switching. Am J Hum Genet. 1993;53:113–24.
-
Luo J-H, Ren B, Keryanov S, Tseng GC, Rao UNM, Monga SP, Strom South, Demetris AJ, Nalesnik Chiliad, Yu YP, Ranganathan S, Michalopoulos GK. Transcriptomic and genomic analysis of human hepatocellular carcinomas and hepatoblastomas. Hepatology. 2006;44:1012–24.
-
de Kok JB, Verhaegh GW, Roelofs RW, Hessels D, Kiemeney LA, Aalders TW, Swinkels DW, Schalken JA. DD3(PCA3), a very sensitive and specific marker to detect prostate tumors. Cancer Res. 2002;62:2695–viii.
-
Yan X, Hu Z, Feng Y, Hu X, Yuan J, Zhao SD, Zhang Y, Yang L, Shan West, He Q, Fan L, Kandalaft LE, Tanyi JL, Li C, Yuan C-X, Zhang D, Yuan H, Hua Thousand, Lu Y, Katsaros D, Huang Q, Montone K, Fan Y, Coukos G, Boyd J, Sood AK, Rebbeck T, Mills GB, Dang CV, Zhang 50. Comprehensive genomic label of long not-coding RNAs across homo cancers. Cancer Cell. 2015;28:529–xl.
-
Clark MB, Mercer TR, Bussotti G, Leonardi T, Haynes KR, Crawford J, Brunck ME, Cao 1000-AL, Thomas GP, Chen WY, Taft RJ, Nielsen LK, Enright AJ, Mattick JS, Dinger ME. Quantitative gene profiling of long noncoding RNAs with targeted RNA sequencing. Nat Meth. 2015;12:339–42.
-
Hu X, Feng Y, Zhang D, Zhao SD, Hu Z, Greshock J, Zhang Y, Yang L, Zhong X, Wang L-P, Jean Southward, Li C, Huang Q, Katsaros D, Montone KT, Tanyi JL, Lu Y, Boyd J, Nathanson KL, Li H, Mills GB, Zhang Fifty. A functional genomic arroyo identifies FAL1 as an oncogenic long noncoding RNA that associates with BMI1 and represses p21 expression in cancer. Cancer Prison cell. 2014;26:344–57.
-
Zhou C-C, Yang F, Yuan South-X, Ma J-Z, Liu F, Yuan J-H, Bi F-R, Lin G-Y, Yin J-H, Cao G-West, Zhou Due west-P, Wang F, Sunday South-H. Systemic genome screening identified the result associated focal loss of long noncoding RNA PRAL in hepatocellular carcinoma. Hepatology. 2015;63:850.
-
Hart JR, Roberts TC, Weinberg MS, Morris KV, Vogt PK. MYC regulates the non-coding transcriptome. Oncotarget. 2014;5:12543–54.
-
Younger ST, Kenzelmann-Broz D, Jung H, Attardi LD, Rinn JL. Integrative genomic assay reveals widespread enhancer regulation by p53 in response to Dna damage. Nucleic Acids Res. 2015;43:4447–62.
-
Grossi Due east, Sanchez Y, Huarte M. Expanding the p53 regulatory network: LncRNAs take up the challenge. Biochim Biophys Acta. 1859;2016:200–viii.
-
Doose Thousand, Haake A, Bernhart SH, Lopez C, Duggimpudi S, Wojciech F, Bergmann AK, Borkhardt A, Burkhardt B, Claviez A, Dimitrova 50, Haas S, Hoell JI, Hummel M, Karsch D, Klapper W, Kleo K, Kretzmer H, Kreuz 1000, Kuppers R, Lawerenz C, Lenze D, Loeffler Yard, Mantovani-Loffler L, Moller P, Ott G, Richter J, Rohde Grand, Rosenstiel P, Rosenwald A, et al. MINCR is a MYC-induced lncRNA able to modulate MYC'south transcriptional network in Burkitt lymphoma cells. Proc Natl Acad Sci U S A. 2015;112:E5261–seventy.
-
Li J, Bian E-B, He Ten-J, Ma C-C, Zong Yard, Wang H-L, Zhao B. Epigenetic repression of long non-coding RNA MEG3 mediated by DNMT1 represses the p53 pathway in gliomas. Int J Oncol. 2016;48:723–33.
-
Pasmant E, Sabbagh A, Masliah-Planchon J, Ortonne N, Laurendeau I, Melin 50, Ferkal S, Hernandez L, Leroy K, Valeyrie-Allanore Fifty, Parfait B, Vidaud D, Bieche I, Lantieri L, Wolkenstein P, Vidaud Grand. Role of noncoding RNA ANRIL in genesis of plexiform neurofibromas in neurofibromatosis type ane. J Natl Cancer Inst. 2011;103:1713–22.
-
Jendrzejewski J, He H, Radomska HS, Li W, Tomsic J, Liyanarachchi S, Davuluri RV, Nagy R, la Chapelle de A. The polymorphism rs944289 predisposes to papillary thyroid carcinoma through a big intergenic noncoding RNA factor of tumor suppressor type. Proc Natl Acad Sci U S A. 2012;109:8646–51.
-
Diskin SJ, Capasso M, Schnepp RW, Cole KA, Attiyeh EF, Hou C, Diamond 1000, Carpenter EL, Wintertime C, Lee H, Jagannathan J, Latorre V, Iolascon A, Hakonarson H, Devoto Chiliad, Maris JM. Common variation at 6q16 inside HACE1 and LIN28B influences susceptibility to neuroblastoma. Nat Genet. 2012;44:1126–30.
-
Pandey GK, Mitra South, Subhash S, Hertwig F, Kanduri M, Mishra Yard, Fransson Due south, Ganeshram A, Mondal T, Bandaru S, Ostensson K, Akyurek LM, Abrahamsson J, Pfeifer S, Larsson E, Shi 50, Peng Z, Fischer Yard, Martinsson T, Hedborg F, Kogner P, Kanduri C. The gamble-associated long noncoding RNA NBAT-ane controls neuroblastoma progression by regulating jail cell proliferation and neuronal differentiation. Cancer Cell. 2014;26:722–37.
-
Russell MR, Penikis A, Oldridge DA, Alvarez-Dominguez JR, McDaniel L, Diamond K, Padovan O, Raman P, Li Y, Wei JS, Zhang Southward, Gnanchandran J, Seeger R, Asgharzadeh S, Khan J, Diskin SJ, Maris JM, Cole KA. CASC15-S Is a Tumor Suppressor lncRNA at the 6p22 Neuroblastoma Susceptibility Locus. Cancer Res. 2015;75:3155–66.
-
Kim T, Cui R, Jeon Y-J, Lee J-H, Lee JH, Sim H, Park JK, Fadda P, Tili E, Nakanishi H, Huh Chiliad-I, Kim S-H, Cho JH, Sung BH, Peng Y, Lee TJ, Luo Z, Sun H-L, Wei H, Alder H, Oh JS, Shim KS, Ko S-B, Croce CM. Long-range interaction and correlation between MYC enhancer and oncogenic long noncoding RNA CARLo-v. Proc Natl Acad Sci U S A. 2014;111:4173–viii.
-
Welter D, MacArthur J, Morales J, Burdett T, Hall P, Junkins H, Klemm A, Flicek P, Manolio T, Hindorff L, Parkinson H. The NHGRI GWAS Catalog, a curated resources of SNP-trait associations. Nucleic Acids Res. 2014;42(Database issue):D1001–6.
-
Quek XC, Thomson DW, Maag JLV, Bartonicek Due north, Signal B, Clark MB, Gloss BS, Dinger ME. lncRNAdb v2.0: expanding the reference database for functional long noncoding RNAs. Nucleic Acids Res. 2015;43(Database issue):D168–73.
-
Davidovich C, Cech TR. The recruitment of chromatin modifiers past long noncoding RNAs: lessons from PRC2. RNA. 2015;21:2007–22.
-
Ringrose L, Ehret H, Paro R. Distinct contributions of histone H3 lysine ix and 27 methylation to locus-specific stability of polycomb complexes. Mol Prison cell. 2004;16:641–53.
-
Kotake Y, Nakagawa T, Kitagawa M, Suzuki S, Liu N, Kitagawa M, Xiong Y. Long non-coding RNA ANRIL is required for the PRC2 recruitment to and silencing of p15(INK4B) tumor suppressor gene. Oncogene. 2011;30:1956–62.
-
Gonzalez I, Munita R, Agirre E, Dittmer TA, Gysling Thousand, Misteli T, Luco RF. A lncRNA regulates culling splicing via institution of a splicing-specific chromatin signature. Nat Struct Mol Biol. 2015;22:370–six.
-
Salameh A, Lee AK, Cardo-Vila Thou, Nunes DN, Efstathiou E, Staquicini FI, Dobroff AS, Marchio Due south, Navone NM, Hosoya H, Lauer RC, Wen S, Salmeron CC, Hoang A, Newsham I, Lima LA, Carraro DM, Oliviero Southward, Kolonin MG, Sidman RL, Exercise M-A, Troncoso P, Logothetis CJ, Brentani RR, Calin GA, Cavenee WK, Dias-Neto Eastward, Pasqualini R, Arap West. PRUNE2 is a human being prostate cancer suppressor regulated past the intronic long noncoding RNA PCA3. Proc Natl Acad Sci U S A. 2015;112:8403–8.
-
Davidovich C, Wang X, Cifuentes-Rojas C, Goodrich KJ, Gooding AR, Lee JT, Cech TR. Toward a consensus on the bounden specificity and promiscuity of PRC2 for RNA. Mol Prison cell. 2015;57:552–eight.
-
Mohammad F, Mondal T, Guseva N, Pandey GK, Kanduri C. Kcnq1ot1 noncoding RNA mediates transcriptional gene silencing by interacting with Dnmt1. Development. 2010;137:2493–9.
-
Li Q, Su Z, Xu X, Liu Yard, Song Ten, Wang R, Sui Ten, Liu T, Chang X, Huang D. AS1DHRS4, a head-to-head natural antisense transcript, silences the DHRS4 gene cluster in cis and trans. Proc Natl Acad Sci U South A. 2012;109:14110–5.
-
Merry CR, Forrest ME, Sabers JN, Bristles L, Gao 10-H, Hatzoglou M, Jackson MW, Wang Z, Markowitz SD, Khalil AM. DNMT1-associated long non-coding RNAs regulate global gene expression and Deoxyribonucleic acid methylation in colon cancer. Hum Mol Genet. 2015;24:6240–53.
-
Lee RS, Roberts CWM. Linking the SWI/SNF circuitous to prostate cancer. Nat Genet. 2013;45:1268–9.
-
Arab 1000, Park YJ, Lindroth AM, Schafer A, Oakes C, Weichenhan D, Lukanova A, Lundin Eastward, Risch A, Meister M, Dienemann H, Dyckhoff G, Herold-Mende C, Grummt I, Niehrs C, Plass C. Long noncoding RNA TARID directs demethylation and activation of the tumor suppressor TCF21 via GADD45A. Mol Cell. 2014;55:604–fourteen.
-
Yang F, Deng X, Ma Westward, Berletch JB, Rabaia N, Wei Thousand, Moore JM, Filippova GN, Xu J, Liu Y, Noble WS, Shendure J, Disteche CM. The lncRNA Firre anchors the inactive X chromosome to the nucleolus past binding CTCF and maintains H3K27me3 methylation. Genome Biol. 2015;xvi:52.
-
Wang KC, Yang YW, Liu B, Sanyal A, Corces-Zimmerman R, Chen Y, Lajoie BR, Protacio A, Flynn RA, Gupta RA, Wysocka J, Lei M, Dekker J, Helms JA, Chang HY. A long noncoding RNA maintains agile chromatin to coordinate homeotic gene expression. Nature. 2011;472:120–4.
-
Lai F, Orom UA, Cesaroni M, Beringer M, Taatjes DJ, Blobel GA, Shiekhattar R. Activating RNAs associate with Mediator to enhance chromatin architecture and transcription. Nature. 2013;494:497–501.
-
Xiang J-F, Yin Q-F, Chen T, Zhang Y, Zhang X-O, Wu Z, Zhang South, Wang H-B, Ge J, Lu X, Yang 50, Chen Fifty-L. Human colorectal cancer-specific CCAT1-L lncRNA regulates long-range chromatin interactions at the MYC locus. Jail cell Res. 2014;24:513–31.
-
Takayama Grand-I, Horie-Inoue K, Katayama Due south, Suzuki T, Tsutsumi Due south, Ikeda 1000, Urano T, Fujimura T, Takagi K, Takahashi Due south, Homma Y, Ouchi Y, Aburatani H, Hayashizaki Y, Inoue S. Androgen-responsive long noncoding RNA CTBP1-AS promotes prostate cancer. EMBO J. 2013;32:1665–fourscore.
-
Ling H, Spizzo R, Atlasi Y, Nicoloso 1000, Shimizu Thousand, Redis RS, Nishida N, Gafa R, Song J, Guo Z, Ivan C, Barbarotto E, De Vries I, Zhang Ten, Ferracin M, Churchman M, van Galen JF, Beverloo BH, Shariati M, Haderk F, Estecio MR, Garcia-Manero G, Patijn GA, Gotley DC, Bhardwaj V, Shureiqi I, Sen Due south, Multani Equally, Welsh J, Yamamoto K, et al. CCAT2, a novel noncoding RNA mapping to 8q24, underlies metastatic progression and chromosomal instability in colon cancer. Genome Res. 2013;23:1446–61.
-
Wang G, Lunardi A, Zhang J, Chen Z, Ala U, Webster KA, Tay Y, Gonzalez-Billalabeitia E, Egia A, Shaffer DR, Carver B, Liu 10-Southward, Taulli R, Kuo WP, Nardella C, Signoretti S, Cordon-Cardo C, Gerald WL, Pandolfi PP. Zbtb7a suppresses prostate cancer through repression of a Sox9-dependent pathway for cellular senescence bypass and tumor invasion. Nat Genet. 2013;45:739–46.
-
Li Y, Wang Z, Shi H, Li H, Li L, Fang R, Cai X, Liu B, Zhang X, Ye L. HBXIP and LSD1 Scaffolded by lncRNA hotair mediate transcriptional activation by c-Myc. Cancer Res. 2015;76:293.
-
Clemson CM, Hutchinson JN, Sara SA, Ensminger AW, Fox AH, Chess A, Lawrence JB. An architectural function for a nuclear noncoding RNA: NEAT1 RNA is essential for the construction of paraspeckles. Mol Cell. 2009;33:717–26.
-
Naganuma T, Nakagawa Due south, Tanigawa A, Sasaki YF, Goshima N, Hirose T. Alternative three′-end processing of long noncoding RNA initiates construction of nuclear paraspeckles. EMBO J. 2012;31:4020–34.
-
Xing Z, Lin A, Li C, Liang K, Wang S, Liu Y, Park PK, Qin 50, Wei Y, Hawke DH, Hung M-C, Lin C, Yang Fifty. lncRNA directs cooperative epigenetic regulation downstream of chemokine signals. Cell. 2014;159:1110–25.
-
Rippe K, Luke B. TERRA and the land of the telomere. Nat Struct Mol Biol. 2015;22:853–viii.
-
Mourtada-Maarabouni K, Pickard MR, Hedge VL, Farzaneh F, Williams GT. GAS5, a non-protein-coding RNA, controls apoptosis and is downregulated in chest cancer. Oncogene. 2009;28:195–208.
-
Tay Y, Rinn J, Pandolfi PP. The multilayered complexity of ceRNA crosstalk and competition. Nature. 2014;505:344–52.
-
Nie W, Ge H-J, Yang 10-Q, Sun X, Huang H, Tao Ten, Chen W-S, Li B. LncRNA-UCA1 exerts oncogenic functions in not-small cell lung cancer by targeting miR-193a-3p. Cancer Lett. 2016;371:99–106.
-
Peng W, Si Due south, Zhang Q, Li C, Zhao F, Wang F, Yu J, Ma R. Long non-coding RNA MEG3 functions equally a competing endogenous RNA to regulate gastric cancer progression. J Exp Clin Cancer Res. 2015;34:79.
-
Deng L, Yang S-B, Xu F-F, Zhang J-H. Long noncoding RNA CCAT1 promotes hepatocellular carcinoma progression by functioning as allow-7 sponge. J Exp Clin Cancer Res. 2015;34:18.
-
Zhou X, Gao Q, Wang J, Zhang X, Liu K, Duan Z. Linc-RNA-RoR acts as a "sponge" against mediation of the differentiation of endometrial cancer stem cells by microRNA-145. Gynecol Oncol. 2014;133:333–9.
-
Wang P, Liu Y-H, Yao Y-50, Li Z, Li Z-Q, Ma J, Xue Y-X. Long non-coding RNA CASC2 suppresses malignancy in human gliomas past miR-21. Cell Signal. 2015;27:275–82.
-
Liu B, Sun L, Liu Q, Gong C, Yao Y, Lv X, Lin L, Yao H, Su F, Li D, Zeng 1000, Song Due east. A cytoplasmic NF-kappaB interacting long noncoding RNA blocks IkappaB phosphorylation and suppresses chest cancer metastasis. Cancer Cell. 2015;27:370–81.
-
Lee S, Kopp F, Chang T-C, Sataluri A, Chen B, Sivakumar Southward, Yu H, Xie Y, Mendell JT. Noncoding RNA NORAD regulates genomic stability past sequestering PUMILIO proteins. Cell. 2015;164:69.
-
Postepska-Igielska A, Giwojna A, Gasri-Plotnitsky 50, Schmitt N, Dold A, Ginsberg D, Grummt I. LncRNA Khps1 regulates expression of the proto-oncogene SPHK1 via triplex-mediated changes in chromatin structure. Mol Jail cell. 2015;lx:626–36.
-
Mondal T, Subhash S, Vaid R, Enroth Due south, Uday S, Reinius B, Mitra S, Mohammed A, James AR, Hoberg E, Moustakas A, Gyllensten U, Jones SJM, Gustafsson CM, Sims AH, Westerlund F, Gorab E, Kanduri C. MEG3 long noncoding RNA regulates the TGF-beta pathway genes through formation of RNA-Deoxyribonucleic acid triplex structures. Nat Commun. 2015;6:7743.
-
Liz J, Portela A, Soler G, Gomez A, Ling H, Michlewski Grand, Calin GA, Guil S, Esteller M. Regulation of pri-miRNA processing by a long noncoding RNA transcribed from an ultraconserved region. Mol Cell. 2014;55:138–47.
-
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57–70.
-
Hanahan D, Weinberg RA. Hallmarks of cancer: the side by side generation. Cell. 2011;144:646–74.
-
Gutschner T, Diederichs South. The hallmarks of cancer: a long non-coding RNA point of view. RNA Biol. 2012;9:703–19.
-
Cathcart P, Lucchesi Westward, Ottaviani Due south, De Giorgio A, Krell J, Stebbing J, Castellano L. Noncoding RNAs and the command of signalling via nuclear receptor regulation in health and affliction. Best Pract Res Clin Endocrinol Metab. 2015;29:529–43.
-
Lanz RB, McKenna NJ, Onate SA, Albrecht U, Wong J, Tsai SY, Tsai MJ, O'Malley BW. A steroid receptor coactivator, SRA, functions equally an RNA and is present in an SRC-i complex. Cell. 1999;97:17–27.
-
Shi Y, Downes M, Xie Westward, Kao HY, Ordentlich P, Tsai CC, Hon Thou, Evans RM. Abrupt, an inducible cofactor that integrates nuclear receptor repression and activation. Genes Dev. 2001;xv:1140–51.
-
Zhou Q, Chen J, Feng J, Wang J. Long noncoding RNA PVT1 modulates thyroid cancer cell proliferation past recruiting EZH2 and regulating thyroid-stimulating hormone receptor (TSHR). Tumour Biol. 2015.
-
Kitagawa One thousand, Kitagawa Grand, Kotake Y, Niida H, Ohhata T. Jail cell bicycle regulation by long non-coding RNAs. Cell Mol Life Sci. 2013;70:4785–94.
-
Puvvula PK, Desetty RD, Pineau P, Marchio A, Moon A, Dejean A, Bischof O. Long noncoding RNA PANDA and scaffold-attachment-factor SAFA control senescence entry and exit. Nat Commun. 2014;5:5323.
-
Benatti P, Belluti Southward, Miotto B, Neusiedler J, Dolfini D, Drac M, Basile V, Schwob East, Mantovani R, Julian Accident J, Imbriano C. Direct non transcriptional role of NF-Y in DNA replication. Biochim Biophys Acta. 2015;1863:673.
-
Dimitrova N, Zamudio JR, Jong RM, Soukup D, Resnick R, Sarma G, Ward AJ, Raj A, Lee JT, Sharp PA, Jacks T. LincRNA-p21 activates p21 in cis to promote Polycomb target factor expression and to enforce the G1/S checkpoint. Mol Prison cell. 2014;54:777–xc.
-
Liu 10, Li D, Zhang Westward, Guo M, Zhan Q. Long not-coding RNA gadd7 interacts with TDP-43 and regulates Cdk6 mRNA decay. EMBO J. 2012;31:4415–27.
-
Poliseno L, Salmena L, Zhang J, Carver B, Haveman WJ, Pandolfi PP. A coding-independent function of cistron and pseudogene mRNAs regulates neoplasm biology. Nature. 2010;465:1033–8.
-
DeOcesano-Pereira C, Amaral MS, Parreira KS, Ayupe Air-conditioning, Jacysyn JF, Amarante-Mendes GP, Reis EM, Verjovski-Almeida S. Long not-coding RNA INXS is a critical mediator of BCL-XS induced apoptosis. Nucleic Acids Res. 2014;42:8343–55.
-
Mazar J, Zhao W, Khalil AM, Lee B, Shelley J, Govindarajan SS, Yamamoto F, Ratnam M, Aftab MN, Collins South, Finck BN, Han 10, Mattick JS, Dinger ME, Perera RJ. The functional characterization of long noncoding RNA SPRY4-IT1 in homo melanoma cells. Oncotarget. 2014;v:8959–69.
-
Zhao H, Zhang Ten, Frazao JB, Condino-Neto A, Newburger PE. HOX antisense lincRNA HOXA-AS2 is an apoptosis repressor in all trans retinoic acid treated NB4 promyelocytic leukemia cells. J Cell Biochem. 2013;114:2375–83.
-
Yang H, Zhong Y, Xie H, Lai X, Xu Thou, Nie Y, Liu S, Wan Y-JY. Consecration of the liver cancer-down-regulated long noncoding RNA uc002mbe.2 mediates trichostatin-induced apoptosis of liver cancer cells. Biochem Pharmacol. 2013;85:1761–9.
-
Redon Due south, Reichenbach P, Lingner J. The not-coding RNA TERRA is a natural ligand and direct inhibitor of man telomerase. Nucleic Acids Res. 2010;38:5797–806.
-
Fu West-M, Lu Y-F, Hu B-One thousand, Liang West-C, Zhu X, Yang H-D, Li G, Zhang J-F. Long noncoding RNA hotair mediated angiogenesis in nasopharyngeal carcinoma by direct and indirect signaling pathways. Oncotarget. 2015;vii:4712.
-
Yan B, Yao J, Liu J-Y, Li X-M, Wang X-Q, Li Y-J, Tao Z-F, Song Y-C, Chen Q, Jiang Q. lncRNA-MIAT regulates microvascular dysfunction by performance as a competing endogenous RNA. Circ Res. 2015;116:1143–56.
-
Yuan S-X, Yang F, Yang Y, Tao Q-F, Zhang J, Huang G, Yang Y, Wang R-Y, Yang S, Huo X-Southward, Zhang L, Wang F, Sun Due south-H, Zhou West-P. Long noncoding RNA associated with microvascular invasion in hepatocellular carcinoma promotes angiogenesis and serves equally a predictor for hepatocellular carcinoma patients' poor recurrence-free survival afterwards hepatectomy. Hepatology. 2012;56:2231–41.
-
Michalik KM, Yous Ten, Manavski Y, Doddaballapur A, Zornig M, Braun T, John D, Ponomareva Y, Chen W, Uchida S, Boon RA, Dimmeler Due south. Long noncoding RNA MALAT1 regulates endothelial jail cell role and vessel growth. Circ Res. 2014;114:1389–97.
-
Raveh E, Matouk IJ, Gilon M, Hochberg A. The H19 Long non-coding RNA in cancer initiation, progression and metastasis - a proposed unifying theory. Mol Cancer. 2015;xiv:184.
-
Yang M-H, Hu Z-Y, Xu C, Xie L-Y, Wang X-Y, Chen S-Y, Li Z-K. MALAT1 promotes colorectal cancer cell proliferation/migration/invasion via PRKA kinase anchor protein nine. Biochim Biophys Acta. 2015;1852:166–74.
-
Arun G, Diermeier S, Akerman M, Chang One thousand-C, Wilkinson JE, Hearn S, Kim Y, MacLeod AR, Krainer AR, Norton L, Brogi E, Egeblad G, Spector DL. Differentiation of mammary tumors and reduction in metastasis upon Malat1 lncRNA loss. Genes Dev. 2016;xxx:34–51.
-
Deng J, Liang Y, Liu C, He S, Wang S. The upwardly-regulation of long non-coding RNA AFAP1-AS1 is associated with the poor prognosis of NSCLC patients. Biomed Pharmacother. 2015;75:eight–11.
-
Qiu M, Xu Y, Yang X, Wang J, Hu J, Xu L, Yin R. CCAT2 is a lung adenocarcinoma-specific long non-coding RNA and promotes invasion of non-small prison cell lung cancer. Neoplasm Biol. 2014;35:5375–80.
-
Eades G, Wolfson B, Zhang Y, Li Q, Yao Y, Zhou Q. lincRNA-RoR and miR-145 regulate invasion in triple-negative breast cancer via targeting ARF6. Mol Cancer Res. 2015;xiii:330–8.
-
Han Y, Ye J, Wu D, Wu P, Chen Z, Chen J, Gao S, Huang J. LEIGC long non-coding RNA acts as a tumor suppressor in gastric carcinoma by inhibiting the epithelial-to-mesenchymal transition. BMC Cancer. 2014;14:932.
-
Yuan J-H, Yang F, Wang F, Ma J-Z, Guo Y-J, Tao Q-F, Liu F, Pan W, Wang T-T, Zhou C-C, Wang S-B, Wang Y-Z, Yang Y, Yang Due north, Zhou Westward-P, Yang G-S, Sun Due south-H. A long noncoding RNA activated by TGF-beta promotes the invasion-metastasis cascade in hepatocellular carcinoma. Cancer Cell. 2014;25:666–81.
-
Gatenby RA, Gillies RJ. Why do cancers have high aerobic glycolysis? Nat Rev Cancer. 2004;4:891–9.
-
Yang F, Zhang H, Mei Y, Wu M. Reciprocal regulation of HIF-1alpha and lincRNA-p21 modulates the Warburg effect. Mol Cell. 2014;53:88–100.
-
Prensner JR, Chen W, Iyer MK, Cao Q, Ma T, Han S, Sahu A, Malik R, Wilder-Romans Yard, Navone N, Logothetis CJ, Araujo JC, Pisters LL, Tewari AK, Canman CE, Knudsen KE, Kitabayashi North, Rubin MA, Demichelis F, Lawrence TS, Chinnaiyan AM, Feng FY. PCAT-1, a long noncoding RNA, regulates BRCA2 and controls homologous recombination in cancer. Cancer Res. 2014;74:1651–60.
-
Sharma V, Khurana S, Kubben N, Abdelmohsen K, Oberdoerffer P, Gorospe Thousand, Misteli T. A BRCA1-interacting lncRNA regulates homologous recombination. EMBO Rep. 2015;sixteen:1520–34.
-
Wang Y, Zhang D, Wu M, Zhao Q, Nie Y, Fan D. Long noncoding RNA MRUL promotes ABCB1 expression in multidrug-resistant gastric cancer cell sublines. Mol Cell Biol. 2014;34:3182–93.
-
Rhoads A, Au KF. PacBio sequencing and its applications. Genomics Proteomics Bioinformatics. 2015;13:278–89.
-
Cabili MN, Dunagin MC, McClanahan PD, Biaesch A, Padovan-Merhar O, Regev A, Rinn JL, Raj A. Localization and abundance analysis of human lncRNAs at unmarried-cell and single-molecule resolution. Genome Biol. 2015;sixteen:twenty.
-
Kertesz M, Wan Y, Mazor E, Rinn JL, Nutter RC, Chang HY, Segal E. Genome-wide measurement of RNA secondary structure in yeast. Nature. 2010;467:103–seven.
-
Underwood JG, Uzilov AV, Katzman S, Onodera CS, Mainzer JE, Mathews DH, Lowe TM, Salama SR, Haussler D. FragSeq: transcriptome-wide RNA structure probing using high-throughput sequencing. Nat Meth. 2010;7:995–1001.
-
Merino EJ, Wilkinson KA, Coughlan JL, Weeks KM. RNA structure analysis at unmarried nucleotide resolution by selective 2′-hydroxyl acylation and primer extension (SHAPE). J Am Chem Soc. 2005;127:4223–31.
-
Meyer KD, Saletore Y, Zumbo P, Elemento O, Mason CE, Jaffrey SR. Comprehensive analysis of mRNA methylation reveals enrichment in three′ UTRs and near cease codons. Jail cell. 2012;149:1635–46.
-
Delatte B, Wang F, Ngoc LV, Collignon E, Bonvin E, Deplus R, Calonne E, Hassabi B, Putmans P, Awe S, Wetzel C, Kreher J, Soin R, Creppe C, Limbach PA, Gueydan C, Kruys V, Brehm A, Minakhina Due south, Defrance M, Steward R, Fuks F. Transcriptome-wide distribution and function of RNA hydroxymethylcytosine. Science. 2016;351:282–v.
-
Suzuki T, Ueda H, Okada S, Sakurai M. Transcriptome-wide identification of adenosine-to-inosine editing using the Ice-seq method. Nat Protoc. 2015;x:715–32.
-
Chu C, Qu K, Zhong FL, Artandi SE, Chang HY. Genomic maps of long noncoding RNA occupancy reveal principles of RNA-chromatin interactions. Mol Cell. 2011;44:667–78.
-
Chu C, Zhang QC, da Rocha ST, Flynn RA, Bharadwaj M, Calabrese JM, Magnuson T, Heard E, Chang HY. Systematic discovery of Xist RNA binding proteins. Jail cell. 2015;161:404–16.
-
Engreitz JM, Pandya-Jones A, McDonel P, Shishkin A, Sirokman K, Surka C, Kadri S, Xing J, Goren A, Lander ES, Plath K, Guttman One thousand. The Xist lncRNA exploits three-dimensional genome architecture to spread across the Ten chromosome. Science. 2013;341:1237973.
-
Simon MD. Capture hybridization analysis of RNA targets (Chart). Curr Protoc Mol Biol. 2013;Chapter 21:Unit 21.25.
-
Quinn JJ, Chang HY. In situ dissection of RNA functional subunits past domain-specific chromatin isolation by RNA purification (dChIRP). Methods Mol Biol. 2015;1262:199–213.
-
Zhang H, Zeitz MJ, Wang H, Niu B, Ge S, Li Westward, Cui J, Wang K, Qian K, Higgins MJ, Fan Ten, Hoffman AR, Hu J-F. Long noncoding RNA-mediated intrachromosomal interactions promote imprinting at the Kcnq1 locus. J Cell Biol. 2014;204:61–75.
-
Khalil AM, Guttman Thou, Huarte Yard, Garber Thou, Raj A, Rivea Morales D, Thomas K, Presser A, Bernstein Be, van Oudenaarden A, Regev A, Lander ES, Rinn JL. Many human large intergenic noncoding RNAs acquaintance with chromatin-modifying complexes and affect gene expression. Proc Natl Acad Sci U S A. 2009;106:11667–72.
-
Niranjanakumari S, Lasda E, Brazas R, Garcia-Blanco MA. Reversible cross-linking combined with immunoprecipitation to study RNA-poly peptide interactions in vivo. Methods. 2002;26:182–90.
-
Ule J, Jensen K, Mele A, Darnell RB. Prune: a method for identifying protein-RNA interaction sites in living cells. Methods. 2005;37:376–86.
-
Licatalosi DD, Mele A, Fak JJ, Ule J, Kayikci M, Chi SW, Clark TA, Schweitzer Air-conditioning, Blume JE, Wang X, Darnell JC, Darnell RB. HITS-Clip yields genome-broad insights into brain alternative RNA processing. Nature. 2008;456:464–9.
-
Konig J, Zarnack Yard, Rot G, Curk T, Kayikci G, Zupan B, Turner DJ, Luscombe NM, Ule J. iCLIP reveals the office of hnRNP particles in splicing at private nucleotide resolution. Nat Struct Mol Biol. 2010;17:909–xv.
-
Martin L, Meier M, Lyons SM, Sit RV, Marzluff WF, Quake SR, Chang HY. Systematic reconstruction of RNA functional motifs with loftier-throughput microfluidics. Nat Meth. 2012;9:1192–4.
-
Buenrostro JD, Araya CL, Chircus LM, Layton CJ, Chang HY, Snyder MP, Greenleaf WJ. Quantitative analysis of RNA-protein interactions on a massively parallel array reveals biophysical and evolutionary landscapes. Nat Biotechnol. 2014;32:562–8.
-
Ran FA, Hsu PD, Wright J, Agarwala V, Scott DA, Zhang F. Genome engineering using the CRISPR-Cas9 system. Nat Protoc. 2013;8:2281–308.
-
Shechner DM, Hacisuleyman E, Younger ST, Rinn JL. Multiplexable, locus-specific targeting of long RNAs with CRISPR-Display. Nat Meth. 2015;12:664–70.
Acknowledgements
We would like to give thanks Daniel Thomson for his input during the making and proofreading of the manuscript.
Authors' contributions
NB and JLVM designed the outline of the paper. JLVM and NB created all the figures. NB, JLVM and MED wrote the manuscript. All authors take read and approved the final version of this manuscript.
Competing interests
The authors declare that they have no competing interests.
Author information
Affiliations
Corresponding author
Additional file
Rights and permissions
Open Access This article is distributed under the terms of the Artistic Eatables Attribution iv.0 International License (http://creativecommons.org/licenses/past/four.0/), which permits unrestricted use, distribution, and reproduction in whatsoever medium, provided you lot requite appropriate credit to the original author(s) and the source, provide a link to the Artistic Eatables license, and bespeak if changes were made. The Creative Eatables Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
Reprints and Permissions
About this article
Cite this article
Bartonicek, North., Maag, J.Fifty.V. & Dinger, M.E. Long noncoding RNAs in cancer: mechanisms of action and technological advancements. Mol Cancer fifteen, 43 (2016). https://doi.org/x.1186/s12943-016-0530-6
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/s12943-016-0530-6
Keywords
- Vascular Endothelial Growth Gene
- Endogenous RNAs
- Multiple Tumour Blazon
- Hydroxymethylcytosine
- lncRNA Locus
Source: https://molecular-cancer.biomedcentral.com/articles/10.1186/s12943-016-0530-6
0 Response to "State of the Art Technologies Used to Explore Long Noncoding Rnas in Cancer Disease"
Post a Comment